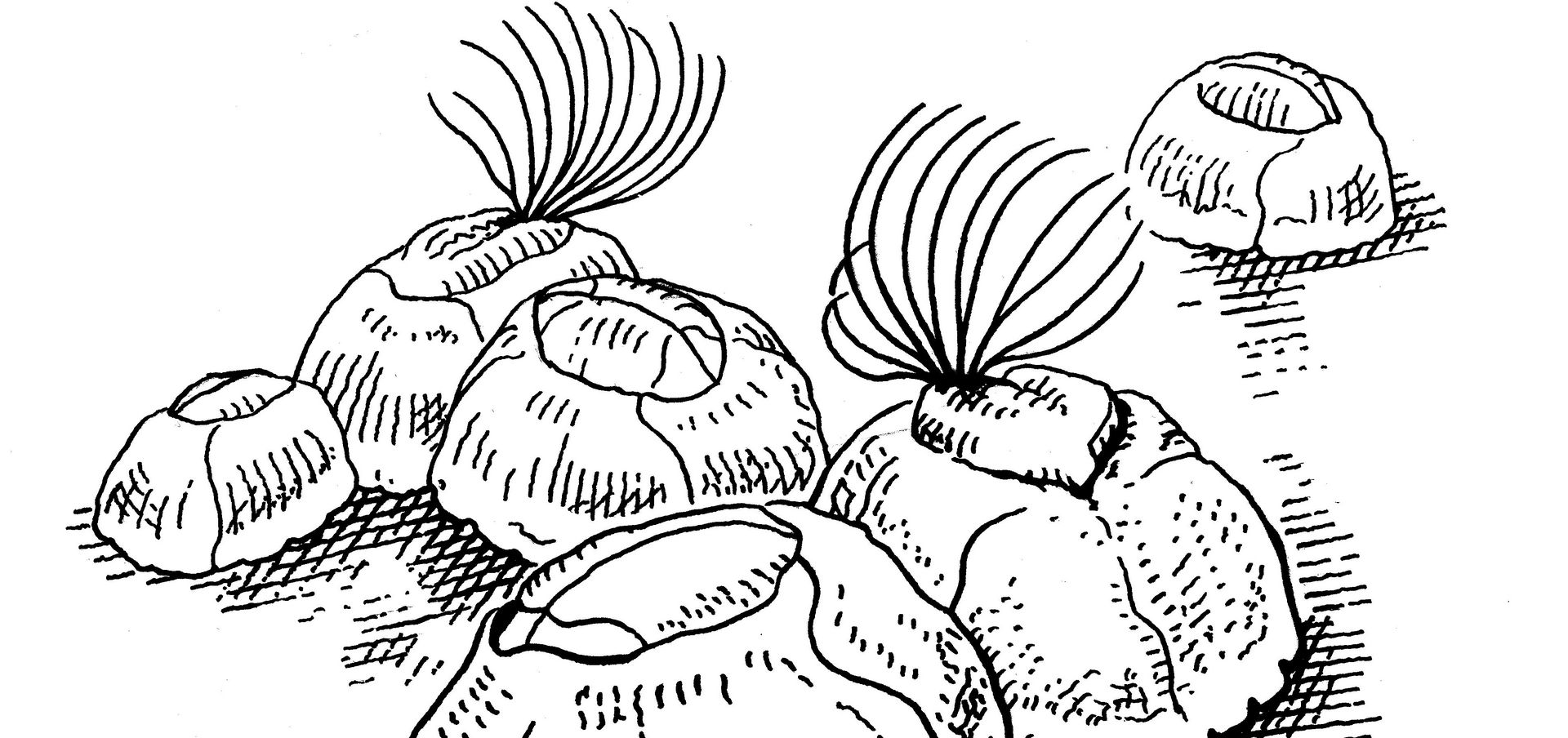
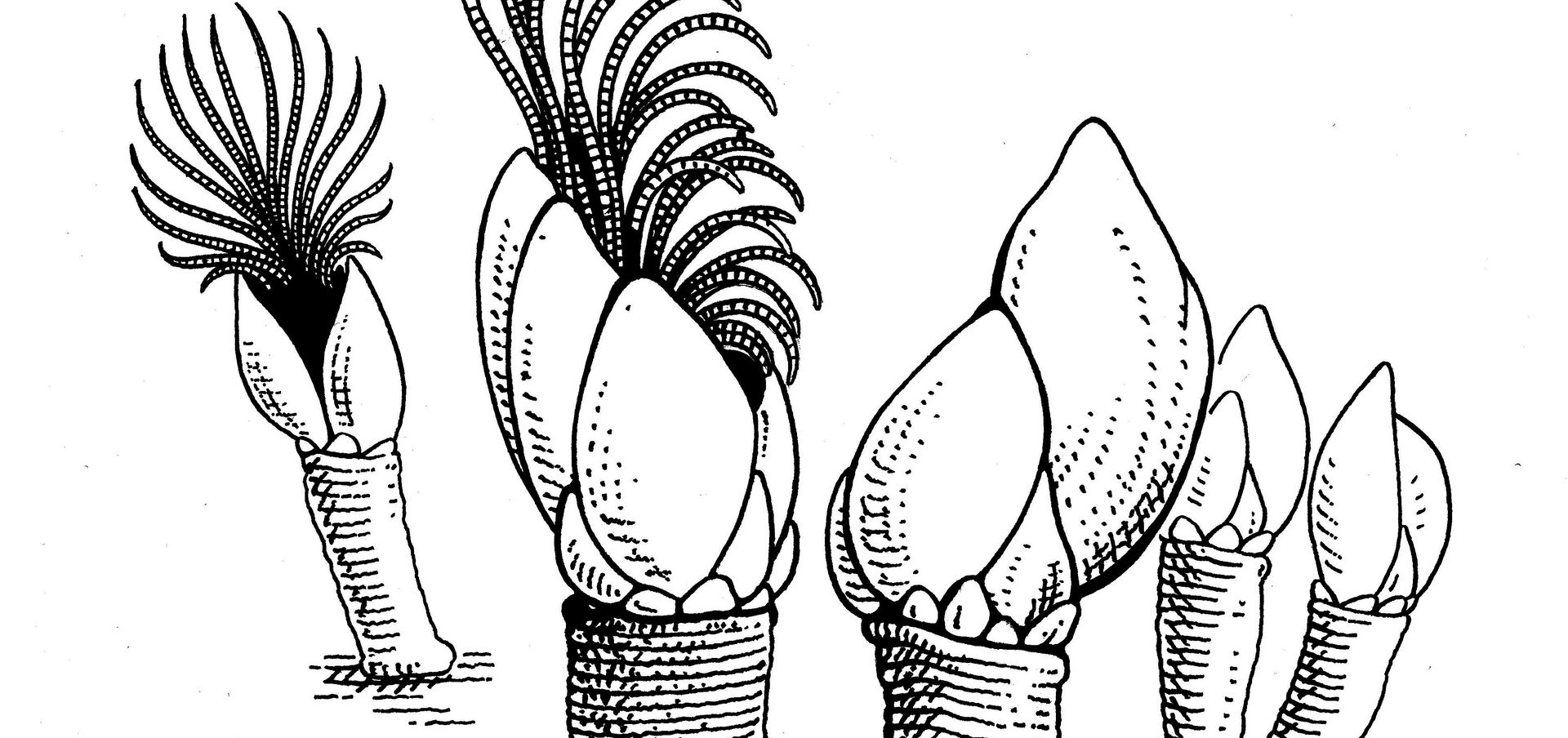
Surfaces exposed to water are colonized by bacteria in next to no time. This bacterial layer provides the perfect gateway community for unicellular algae, fungi and other tiny organisms, which accumulate to form a biofilm. Next come the sessile crustaceans such as goose barnacles, which attach themselves firmly to the biofilm together with algae, sea anemones and soft coral species. This steady proliferation of organisms is known as biofouling, and it causes enough frictional drag to make ships burn significantly more fuel. What’s more, the adhesion of microorganisms leads to a biocorrosion process that can decrease the strength of the ship’s hull or even cause cracks that require costly repairs. Biofouling is a problem that affects not just ships, but also other structures that are permanently situated in water such as bridge piers, wharfs, buoys and offshore rigs. Experts put the cost of downtime and increased maintenance due to biofouling at some 200 billion US dollars a year in the shipping industry alone.
Non-toxic patterning
Our research aims to find an eco-friendly method of preventing biofouling from occurring in the first place. Currently, the shipping industry’s go-to solution involves biocides such as cuprous oxide and zinc pyrithione – substances that exhibit poor biodegradability and pose a major threat to marine organisms. Non-toxic antifouling coatings gradually wear off, causing another form of pollution that affects aquatic habitats. They have now been banned by the US Environmental Protection Agency (EPA).
Our approach is different. Essentially it involves modifying surface structures on a micro and nano scale to prevent microorganisms from attaching themselves to the surface in the first place. It is based on two well-known examples from nature: shark skin and lotus leaves. Lotus leaves are known for their self-cleaning properties. They possess a superhydrophobic surface that causes droplets to “bead” and roll off the leaf, carrying any contaminants with them. This mechanism provides a useful starting point for developing antifouling surfaces. Shark skin has typically attracted researchers’ interest due to its useful hydrodynamic properties, but in this context, we are primarily interested in its surface topographies that prevent microorganism attachment.
Analyzing droplet
Armed with a clear objective and our two examples from nature, we set to work. We began by investigating how microscale topography affects a surface’s hydrophobicity. To do this, we applied a microscale surface pattern using picosecond laser micro-machining. Our aim was to predict the antifouling efficacy of surface structures by comparing the results obtained with a range of different patterns. To classify the rate of microorganism adhesion, we applied two key concepts: engineered roughness index (ERI), and contact angle. The engineered roughness index is designed to illustrate the correlation between the settlement of microorganisms and the wettability of micro-patterned surface topographies. The ERI is a dimensionless value which is calculated as the ratio of the product of Wenzel’s roughness factor (r) and the degree of freedom of the pattern (df) to the depressed surface area fraction (fD).
The other key concept is the contact angle. For our purposes, this is the angle that a water droplet on a surface forms to this surface. This angle characterizes the hydrophobic or hydrophilic nature of the surface. The higher the hydrophobicity, the lower the rate of microorganism adhesion. A surface is hydrophobic if the contact angle is between 90 and 180 degrees, in which case the water droplet does not sit relatively flat on the surface, but instead looks more like a balloon. If the contact angle exceeds 150 degrees, we refer to it as a superhydrophobic surface – one example of which is the lotus leaf.
Anti-Fouling Topography
Using laser pulses, we fabricated patterns on a surface made of poly methyl methacrylate, also known as PMMA. We then studied the patterns under a scanning electron microscope to check the width of the cut and the spacing between adjacent surface features. The results showed that we could achieve the desired properties by creating a circular pattern with two ‘hatch levels’ – microscale square pillars and interconnecting depressions – with a feature size of 100-200 microns. This topological structure leads to a dramatic shift in hydrophobicity. It increases the contact angle of a five milliliter water droplet from 70 degrees for a smooth surface to 113 degrees for the topographically modified surface. At the same time, the ERI value of the surface increases from zero to 1.13. This implies that the rate of microorganism attachment would also drop significantly – and that makes our discovery an effective antifouling solution.
Our plans for the future include experimenting with new patterns and engineered features on different material surfaces to identify the most suitable patterns for real world applications. Making the best choice ultimately depends on finding an economical means of fabricating these micropatterns across the entire expanse of a ship’s hull.

SWAGATIKA PATRA carries out research at Lamar University in Texas along with her research partner Raghabendra Rout under the supervision of Prof. Xinyu Liu. Their goal is to use ultrashort pulse lasers to create non-toxic methods of combating biofouling.